Who Must Register List And Pay The Fee
For the fiscal year of of 2022, the Food and Drug Assistants (FDA) medical device registration fee rates increased by 2.five% from 2021. Considering it increased 6% from 2019 to 2020, this is good news for medical device companies. In FY 2022, which goes from October 1, 2021, to September 30, 2022, that fee is $5,672. In the previous year, it was $5,546. Predicting the toll of registering your medical device can be tough, as the fees change from year to year, and increases aren't always steady. Let's take a look at fee changes from the past decade so yous tin be best prepared for FY 2022 and the years to come. The FDA medical device user fee for 2022 is $five,672. Nevertheless, there are other application fees medical device manufacturers should be aware of when they're working on their budgets. All fees increased by two.v% for 2022. The FDA publishes comprehensive guidelines here, only in general, any company that is involved in the production and distribution of medical devices that are intended to be sold commercially in the U.S. must pay the almanac fee — including devices that are intended for export only. Additionally, most companies that are required to register must besides list the devices and the activities performed on those devices as outlined in Title 21 CFR Part 807. This is different for pocket-size business fees, according to the FDA's site. For all small business submissions, except a 30-twenty-four hours detect and a 513 (g) request, the fee is 25% of the standard fee. For xxx-twenty-four hour period notices and 513 (g) requests, the fee is l% of the standard. Small business organisation conclusion is based on the total amount of gross receipts and sales. If revenue is less than $100,000,000 for the previous fiscal year, and then a business can be eligible. They may as well be eligible for a get-go premarket application waiver if gross receipts and sales are no more than than $thirty,000,000. When we expect back at the Almanac Establishment Registration Fees from 2010 to 2020, we can see that: If we utilize that information to estimate the fees for 2026, then y'all should expect to pay somewhere around $7,715—simply make certain you get out some wiggle room only in instance the corporeality increases more than what's anticipated. If you program to register your device by September 30, 2022—the stop of FY 2022—so you have to pay the FY 2022 registration fee. This fee applies to all medical devices registered between October 1, 2021, and September 30, 2022. Paying the fee is only the kickoff step in the process. Registration isn't complete until y'all receive confirmation from the FDA. Give yourself time to pay the fee, as payment confirmation times can vary based on payment type: If you have questions during the registration process for your medical device, the FDA provides online resource to help, but here are some of the commonly asked questions according to the FDA User Fees Help website. This site gives the information you demand to complete your registration, but if y'all still demand assistance, you lot tin can contact the FDA via e-mail at reglist@cdrh.fda.gov or telephone call them at 301-796-7400. There are iii status types for your PIN: entered, booked, or canceled. You can't alter the fees you have to pay the FDA, but you can salve money by non paying fees for compliance problems. You lot tin learn from the mistakes that other companies make that consequence in warning letters and compliance issues. These include: When the FDA problems a Class 483 violation, yous are responsible for fixing the issue. If you don't, y'all'll get a alarm letter and could face seizure, injunction, or prosecution and fines. One way to assist avert common violations is by using a quality management system, where you can automate tasks, track manufacturing processes, and monitor your pattern history files all in one place. Learn more in: 5 Mutual Medical Device Regulatory Compliance Problems From conception to registration, Qualio tin can help throughout every step of the procedure of building and launching a medical device. Our quality management system was designed with life science companies in mind and can step in to automate tasks, generate reports, and then much more. In addition, we also offer Qualio Plus and Plus Audit services, which requite you direct access to regulatory experts who tin can assist yous throughout the process. Contact us today to see Qualio in action. How Much Is the FDA Medical Device Registration Fee in 2022?
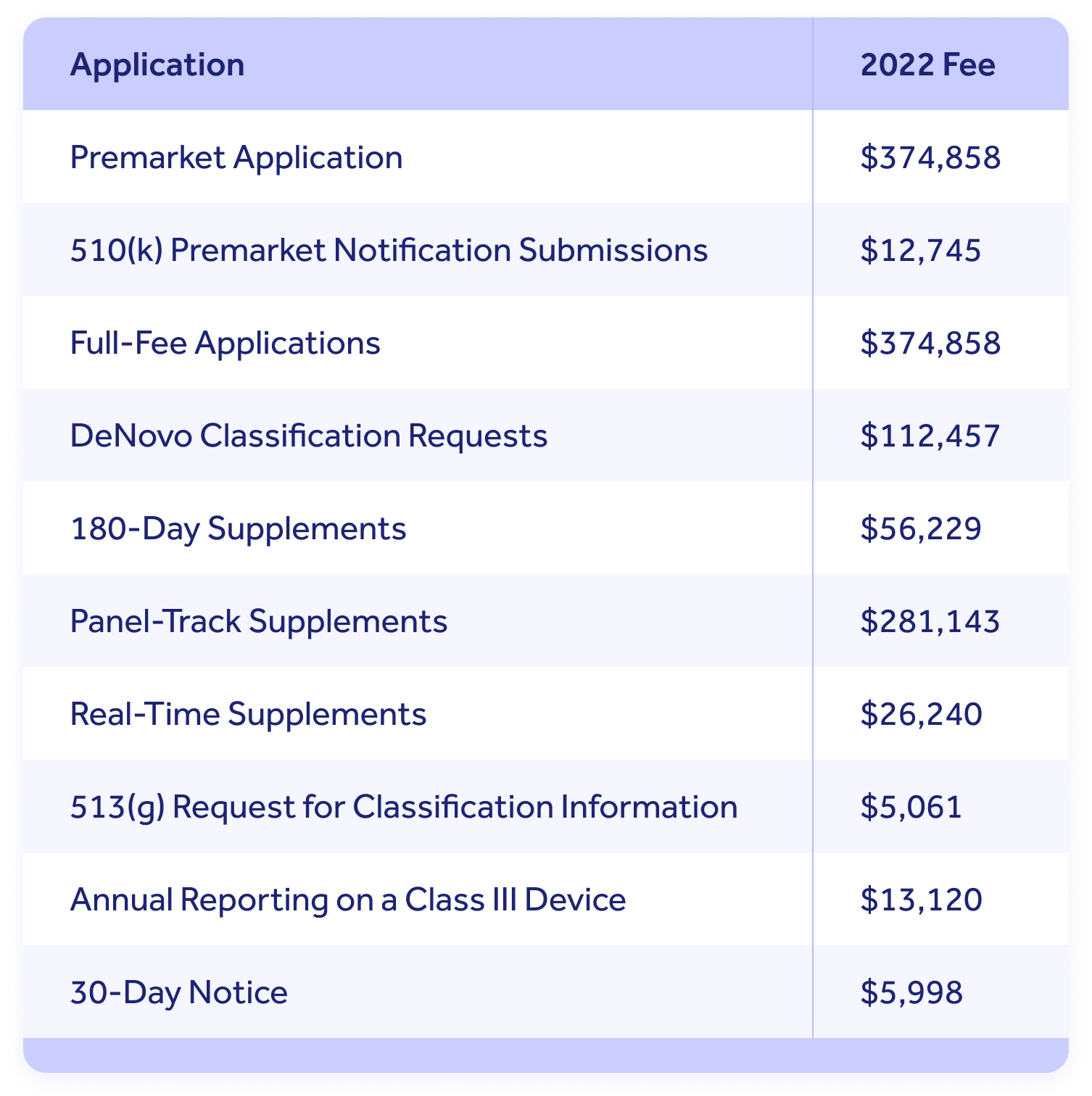
Who Must Register, List, and Pay the Annual Registration Fee?Program Alee: Approximated Annual Establishment Registration Fees
What Is the Borderline for Registering Medical Devices with the FDA?
FDA Medical Device Registration Details
What are the principal steps in the registration procedure?
How can yous get aid during the registration process?
My PIN says it's in "entered" status. What does that hateful?
Save Money by Avoiding Noncompliance Fees
Make all of your medical device creation processes easier with Qualio
Who Must Register List And Pay The Fee,
Source: https://www.qualio.com/blog/fda-medical-device-registration-fee
Posted by: moteolde1960.blogspot.com


0 Response to "Who Must Register List And Pay The Fee"
Post a Comment